October
16, 2007
In
Diabetes, a Complex of Causes
By AMANDA SCHAFFER
An explosion of new research is
vastly changing scientists’ understanding of diabetes and giving new clues about how to
attack it.
The fifth leading killer of
Americans, with 73,000 deaths a year, diabetes is a disease in which the body’s
failure to regulate glucose, or blood sugar, can lead to serious and even fatal
complications. Until very recently, the regulation of glucose — how much
sugar is present in a person’s blood, how much is taken up by cells for fuel,
and how much is released from energy stores — was regarded as a
conversation between a few key players: the pancreas, the liver, muscle and
fat.
Now, however, the party is proving to
be much louder and more complex than anyone had shown before.
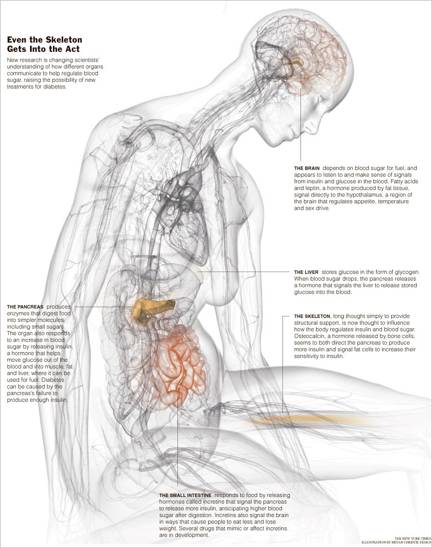
New research suggests that a hormone
from the skeleton, of all places, may influence how the body handles sugar.
Mounting evidence also demonstrates that signals from the immune system, the
brain and the gut play critical roles in controlling glucose and lipid
metabolism. (The findings are mainly relevant to Type 2 diabetes, the more
common kind, which comes on in adulthood.)
Focusing on the cross-talk between
more different organs, cells and molecules represents a “very important change
in our paradigm” for understanding how the body handles glucose, said Dr. C.
Ronald Kahn, a diabetes researcher and professor at Harvard Medical School.
The defining feature of diabetes is
elevated blood sugar. But the reasons for abnormal sugar seem to “differ
tremendously from person to person,” said Dr. Robert A. Rizza, a professor at
the Mayo Clinic College of Medicine. Understanding
exactly what signals are involved, he said, raises the hope of “providing the
right care for each person each day, rather than giving everyone the same
drug.”
Last summer, researchers at Columbia University Medical Center published
startling results showing that a hormone released from bone may help regulate
blood glucose.
When the lead researcher, Dr. Gerard
Karsenty, first described the findings at a conference, the assembled
scientists “were overwhelmed by the potential implications,” said Dr. Saul
Malozowski, senior adviser for endocrine physiology research at the National
Institute of Diabetes and Digestive and Kidney Diseases, who was not involved
in the research. “It was coming from left field. People thought, ‘Oof, this is
really new.’
“For the first time,” he went on, “we
see that the skeleton is actually an endocrine organ,” producing hormones that
act outside of bone.
In previous work, Dr. Karsenty had
shown that leptin, a hormone produced by fat, is an important regulator of bone
metabolism. In this work, he tested the idea that the conversation was a
two-way street. “We hypothesized that if fat regulates bone, bone in essence
must regulate fat,” he said.
Working with mice, he found that a
previously known substance called osteocalcin, which is produced by bone, acted
by signaling fat cells as well as the pancreas. The net effect is to improve
how mice secrete and handle insulin, the hormone that helps the body move
glucose from the bloodstream into cells of the muscle and liver, where it can
be used for energy or stored for future use. Insulin is also important in
regulating lipids.
In Type 2 diabetes, patients’ bodies
no longer heed the hormone’s directives. Their cells are insulin-resistant, and
blood glucose levels surge. Eventually, production of insulin in the pancreas
declines as well.
Dr. Karsenty found that in mice prone
to Type 2 diabetes, an increase in osteocalcin addressed the twin problems of
insulin resistance and low insulin production. That is, it made the mice more
sensitive to insulin and it increased their insulin production, thus bringing
their blood sugar down. As a bonus, it also made obese mice less fat.
If osteocalcin works similarly in
humans, it could turn out to be a “unique new treatment” for Type 2 diabetes,
Dr. Malozowski said. (Most current diabetes drugs either raise insulin
production or improve insulin sensitivity, but not both. Drugs that increase
production tend to make insulin resistance worse.)
A deficiency in osteocalcin could
also turn out to be a cause of Type 2 diabetes, Dr. Karsenty said. Another
recent suspect in glucose regulation is the immune system. In 2003, researchers
from two laboratories found that fat tissue from obese mice contained an
abnormally large number of macrophages, immune cells that contribute to
inflammation. The finding piqued the curiosity of researchers. “I remember
reading the paper and thinking: ‘Wow, look at all those macrophages. What are
they doing?’” said Dr. Jerrold M. Olefsky of the University of California, San Diego, School of
Medicine.
Scientists have long suspected that
inflammation was somehow related to insulin resistance, which precedes nearly
all cases of Type 2 diabetes. In the early 1900s, diabetics were sometimes
given high doses of aspirin, which is an anti-inflammatory, Dr. Olefsky said.
Only in the past few years has
research into the relationship of obesity, inflammation and insulin resistance
become “really hot,” said Dr. Alan R. Saltiel, director of the Life Sciences
Institute at the University of Michigan.
Many researchers agree that obesity
is accompanied by a state of chronic, low-grade inflammation in which some
immune cells are activated, and that that may be a primary cause of insulin
resistance. They also agree that the main type of cell responsible for the
inflammation is the macrophage, Dr. Saltiel said.
But major questions remain, he said:
“Why are these macrophages attracted to fat, liver and muscle in the first
place? What are they doing? What are they secreting? What other immune cells
are in there?”
New research also suggests that “not
all macrophages are created equal,” added Dr. Saltiel. There appear to be “good
ones and bad ones” competing in fat tissue, with potentially large consequences
for inflammation and diabetes.
Meanwhile, the promise of
anti-inflammatory compounds as treatment continues to attract attention.
“Certain cellular anti-inflammatory proteins may now be important new targets
for drug discovery for diabetes treatment,” Dr. Olefsky said. But damping down
the immune system is also potentially risky, he noted, adding: “If you’re
inhibiting the macrophage inflammatory pathway, that’s good for insulin
resistance and diabetes. But it might not be so good for your susceptibility to
infections.” A major goal is to develop a drug that quashes only the specific
component of macrophage inflammation that leads to insulin resistance, without
causing other side effects.
One class of current medications, called
thiazolidinediones, may work in part by reducing inflammation, which may in
turn improve insulin sensitivity. But an example from this class, the drug Avandia, was also found to increase the risk
of heart attacks.
Another participant in the glucose
conversation is the brain. Its role has long been suspected. More than a
century ago, the French physiologist Claude Bernard suggested that the brain
was important in blood sugar regulation. He punctured the brains of
experimental animals in specific areas and managed to derange their blood sugar
metabolism, making them diabetic.
But for years, virtually no one
followed up on this finding, said Dr. Kahn, of Harvard.
People thought about glucose as a
critical fuel for the brain, Dr. Kahn said, but did not explore the brain’s
role in glucose regulation.
Only recently, with more advanced
laboratory techniques, has this role been definitively established and expanded
upon.
Today’s genetic techniques, said Dr.
Rizza, at the Mayo Clinic, are what have “really driven the process.”
For instance, once scientists
developed the ability to manipulate mice so that they lacked particular
receptors in specific tissues, they could show that mice without insulin
receptors in the brain could not regulate glucose properly and went on to
develop diabetes, said Dr. Kahn, whose laboratory published this groundbreaking
work in 2000.
Other researchers have shown that
free fatty acids, as well as the hormone leptin, produced by fat tissue, signal
directly to a part of the brain called the hypothalamus, which also regulates
appetite, temperature and sex drive.
And several recent papers suggest
that direct signaling by glucose itself to neurons in the hypothalamus is also
crucial to normal blood sugar regulation in mice.
“If the brain is getting the message
that you have adequate amounts of these hormones and nutrients, it will
constrain glucose production by the liver and keep blood glucose relatively
low,” said Dr. Michael W. Schwartz, a professor at the University of Washington. But if the brain
senses inadequate amounts, he continued, it will “activate responses that cause
the liver to make more glucose, and new evidence suggests that this contributes
to diabetes and impaired glucose metabolism.”
The brain, therefore, appears to be
listening to — and weighing and making sense of — a chorus of
signals from insulin, leptin, free fatty acids and glucose itself. In response,
it appears to send signals to liver and muscle cells by way of several nerves,
though additional mechanisms are probably involved. The gut also seems to chime
in, said Dr. Rizza, adding that for him, this aspect of sugar regulation came
as “the biggest gee whiz of all.”
“Food comes in through the gut, so of
course you should look there” for molecules involved in glucose regulation, he
said. “But few people realized this until very recently.”
Hormones from the small intestine
called incretins turn out to talk directly with the brain and pancreas in ways
that help reduce blood sugar and cause animals and people to eat less and lose
weight, Dr. Rizza said.
Numerous molecules that mimic
incretins or prevent them from being degraded are in clinical trials. Two such
drugs have been approved by the Food and Drug Administration: Byetta, an
incretin mimic, from Amylin Pharmaceuticals and Eli Lilly; and Januvia, from
Merck, which inhibits the destruction of the incretin GLP1. (Dr. Rizza is an
adviser to Merck but says all consulting fees go to the Mayo Clinic for
education and research.)
Still, it can be hard to predict how
different drugs will interact in the body. And many promising candidates will
turn out to have side effects — chattering helpfully with one organ, but
problematically with another.
“The picture is becoming more and
more complicated,” Dr. Saltiel said. “And let’s face it, it was pretty
complicated before.”
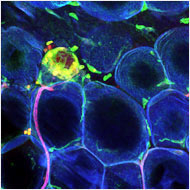
Study Fat tissue from a
mouse that was fed a high-fat diet.
http://www.nytimes.com/2007/10/16/health/16diab.html?%20em&ex=1192593600&en=a89e391b2758f41e&ei=5070